 There are 3 electron regions in the same plane with predicted bond angle of slightly less than 120 ◦. If the atom has 2 sigma bonds and a lone pair of electrons, it is still sp 2 hybridized. An example would be AlBr 3.īent Molecular Geometry, Trigonal Planar Electron Geometry. The orbitals are sp 2 hybridized, the geometry is trigonal planar. When a central atom is involved with 3 sigma bonds, the orbitals will align themselves as far apart as possible around the central nucleus as illustrated here. Trigonal Planar Molecular Geometry, Example Aluminum Bromide See the video and blog "Sigma and Pi Bonds" to see how these orbitals form a triple bond.Ī sigma bond occurs when the electron overlap occurs between the nuclei of the atoms.Ī double bond is a sigma bond and a pi bond.Ī triple bond is a sigma bond and 2 pi bonds. The sp hybridized orbitals, represented by the green are centered and perpendicular to the plane of the yellow p orbitals.Įach carbon in the molecule acetylene can be represented by the electronic orbital configuration of this model. The empty p orbitals, represented by yellow, are perpendicular to each other in the same plane. Now the play dough orbitals are assembled. They will have a linear arrangement, a 180 ◦ bond angle. These two sp hybridized orbitals will orient themselves so that they will be as far apart as possible. Here the blue s and yellow p x orbitals were mixed to form 2 green hybridi zed sp orbitals. The s orbital is represented by the blue sphere and the p orbitals are represented by the yellow dumbbell shapes. Here colored play dough is used to create a 3 dimensional representation of these orbitals. 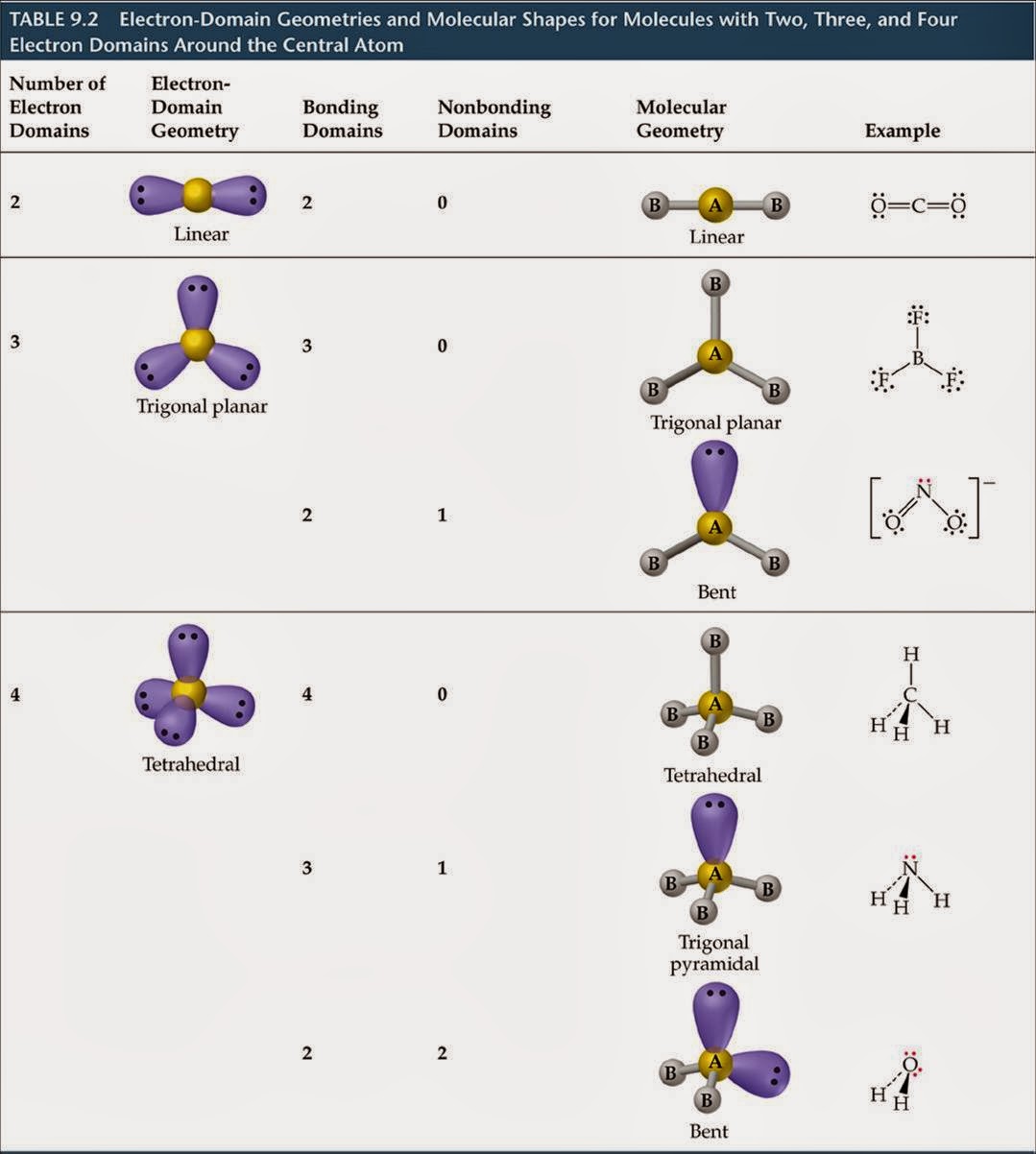
In this illustration they are designated p x, p y, and p z. There are 3 orientations of the p orbital. In this illustration, the probability of finding an electron in the p y orbital is defined by a dumbbell shape with the bulbs of the dumbbell above and below the central atom. In a p orbital the electron can be found on opposite sides of the central nucleus. In an s orbital the electron can be found in a sphere surrounding the nucleus. These shapes define the probability of finding an electron within that space. The geometry is linear.Īn s orbital is in the shape of a sphere and a p orbital is in the shape of a dumbbell. 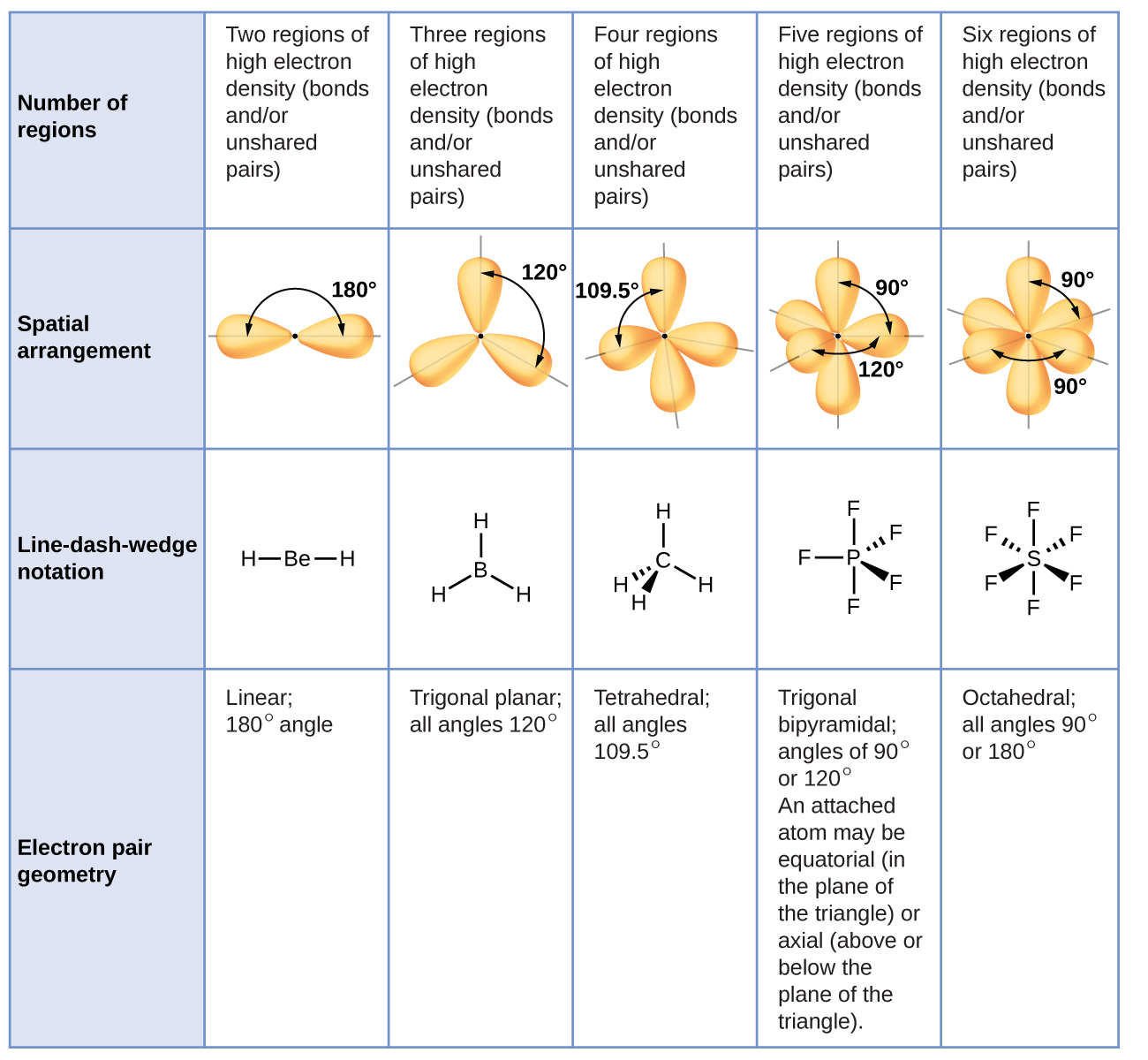
This illustration shows 2 sp hybridized orbitals getting as far apart as possible. This central atom is said to be sp hybridized. If a central atom has 2 sigma ‘ s’ bonds and no lone pairs then the molecular shape is linear with a predicted 180 ◦ bond angle, in other words the orbitals are arranged in a straight line with the central atom. Review the notes after viewing the video: Connect to Wi-Fi to prevent cellular data charges for video streaming. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
